Summary

We are researching stomatal density on different tiers of leaves in Mahonia Aquifolium, also known as Oregon grape. This research will be able to tell us if Oregon grapes adapts to have more or less stomata when needed. The higher the amount of stomata means more CO2 intake and photosynthesis. We know that the higher tiers of Oregon grape cover the lower tiers; and because of this we also know that the lower tiers see less light than the ones above them. Now, our question is, will the higher tiers of leaves have more or less stomatal density than those below it? By establishing an experiment with Oregon grape we will be able to analyze stomatal density vs. tier to create a bar graph showing our data and results.
Intro / Project Background
We hypothesize that the stomatal density of leaves in Mahonia Aquifolium will be greater in the leaves higher up from the ground opposed to the ones lower to

the ground. We predict there will be more stomata on the higher leaves because they see significantly more sun than the underlying leaves. We believe that the higher up leaves seen more sun, therefore have a higher light intake. With a higher light intake we think that the plant needs more stomata on those leaves to be able to conduct photosynthesis more efficiently. According to Keith Mott and Odette Michaelson (Jan. 1991), more light means there will be a higher rate of photosynthesis. Although when comparing stomatal density to light intake they didn’t see any significant change between the three different levels of light. To
counteract this, H. P. Gay and R. G. Hurd found that in a tomato plant there was a higher stomatal density on the top tier leaves; and a lower stomatal density on the leaves closer to the ground. Although there was some variation within the levels of the different leaves, overall the pattern showed a significant difference between the leaves closest to the ground and the ones highest up. In our research, we use the light of the sun, also known as white light. In a study done by Andrew O’Carrigan, Mohammad Babla, Feifei Wang, Xiaohui Liu, Michelle Mak, Richard Thomas, Bill Bellotti, and Zhong-Hua Chen, they conducted a few different experiments with different colors of light to see the response plants had and in what way. They found that in blue light, the stomata was much bigger than it was in red or green light. In our research we will be doing two different comparisons of data. The first set of we collect the tiers of leaves will be as close together as possible. This is data 1-9 and 20-24. The second set of data we collect, the tiers will be from the highest and lowest point on the plant as well as one tier from the middle.This is data 10-19 and 25-30. With some variation in data, we hope to see a difference overall in stomatal density from the higher up leaves rather than the lower. We hypothesize there will be a larger difference in stomatal density when there is a more drastic height difference of each tier.
Methods
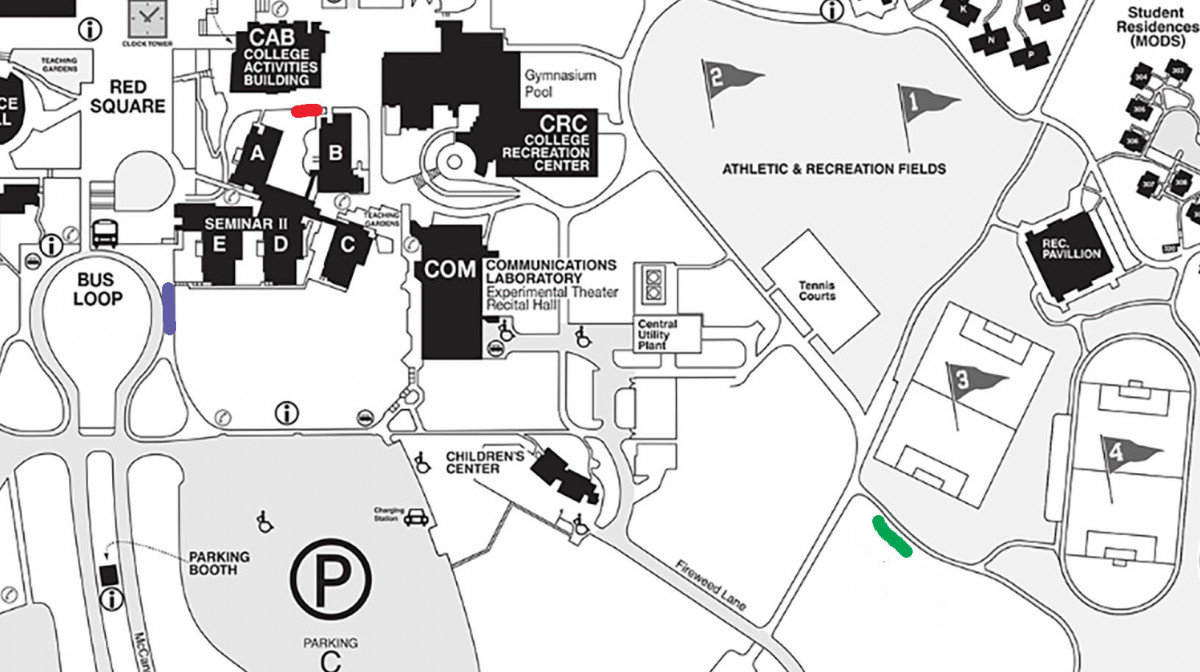
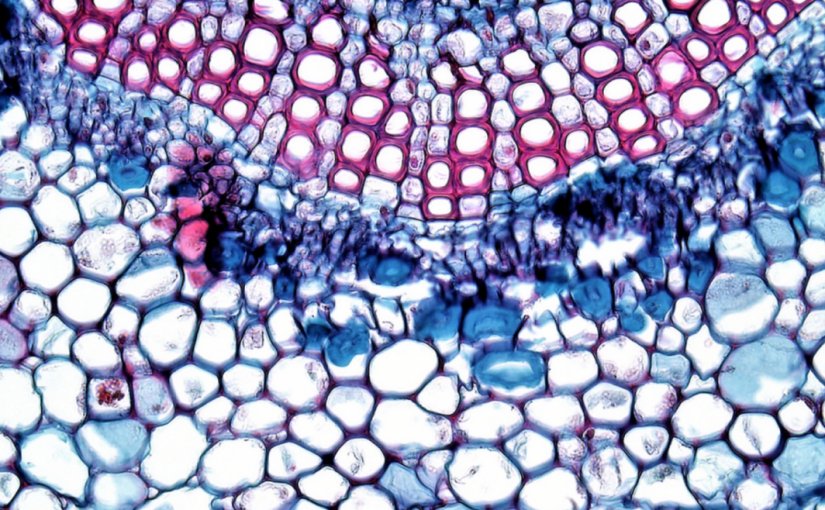
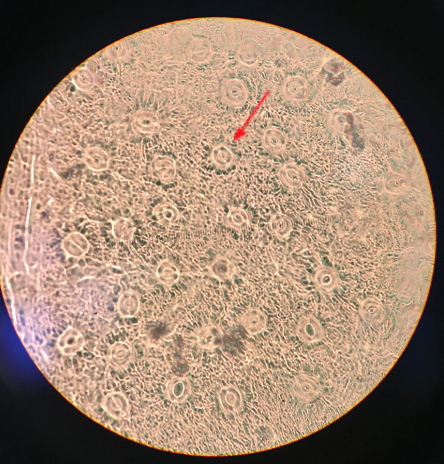
The map below shows where on The Evergreen State campus we are collecting our samples from. We gathered two samples from each of the three tiers of 30 different Oregon grape plants. Samples were collected from the middle of each sporophyte. The three tiers from each sample from samples 1-9 and 20-24 were tiers directly below or above each other, with no other sporophytes in between. For samples 10-19 and 25-30, tier one was the highest on the plant, tier two was in the middle and tier three was the lowest sporophyte of each plant. After collecting all of our 180 samples we brought them to the lab to analyze. By painting each tip of the leaf with a layer of clear nail polish (see in picture to right) then removing it with clear tape, we were able to determine the amount of stomata on each leaf. Video example. After getting a slide prepared and ready to view under the compound microscope, we used a 400x magnification and were able to count the stomata. Microscope setup instructions. The picture to the left is a microscopic image of a leaf from Mahonia Aquifolium. The picture shows us the stomata and with that we are able to tell if they are open or closed, as well as the amount of stomata. In this exact picture, we estimated about 32 stomata. After we got the number of stomata then found the stomatal density by dividing the number of stomata found by the area seen under the microscope. Bruce Grant and Itzick Vatnick do a great job of explaining all the calculations used in this experiment with step by step instructions in their article, Environmental Correlates of Leaf Stomata Density.
Red Plot = Samples 20-24 Purple Plot = Samples 5-14, 25-30 Green Plot = Samples 1-4
Results
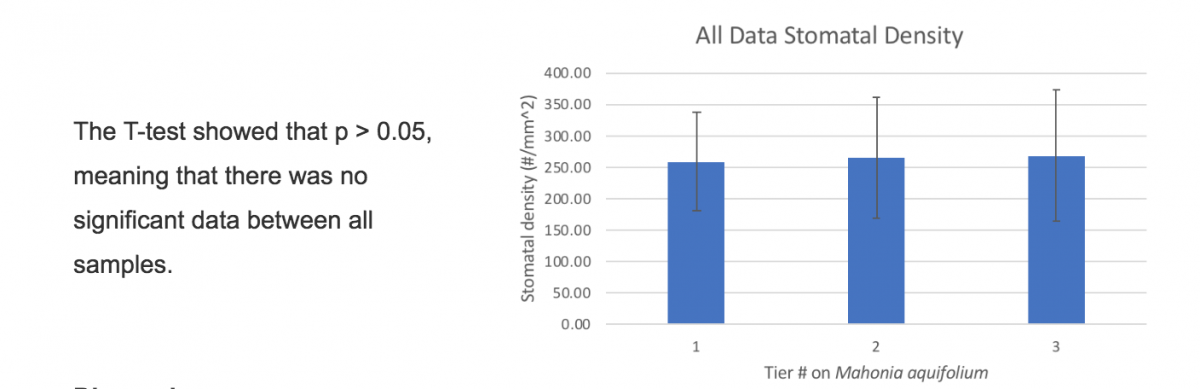
Our data found did not prove or disprove our hypothesis. Although we did not find what we were expecting, we found that there was no overall difference in data. The number of stomata on each leaf, throughout different heights on the plant had no significant difference from one another, meaning no change. Any outliers over 100 stomata per field of view were taken out of our data. Below is a data set for the stomatal density of leaves taken from different tiers of Mahonia Aquifolium (Oregon Grape). Data taken from Evergreen State College and Tumwater Falls (highlighted on map above) spring of 2018. The stomata were counted at 400x, the estimated area of the field of view was calculated to be .1589 mm^2. 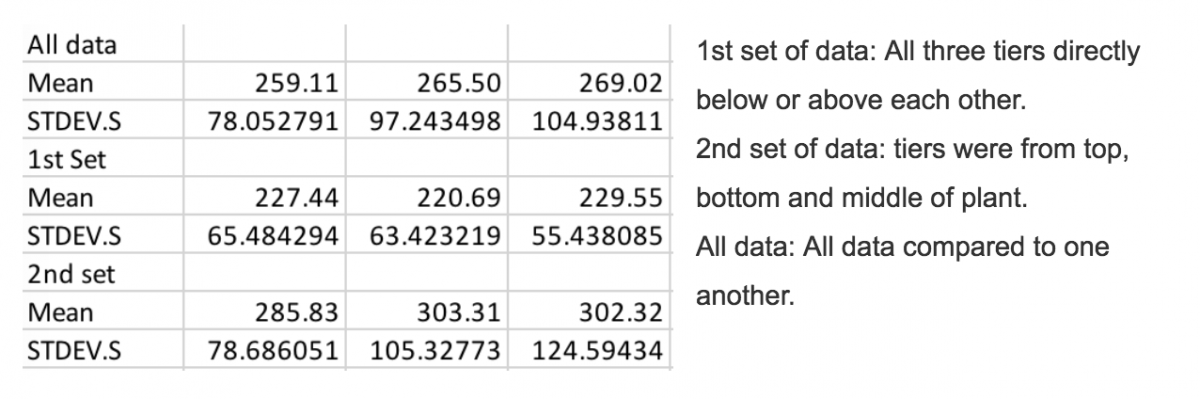
Discussion
In this study, the height of the leaves on Mahonia Aquifolium did not affect the stomatal density.
We hypothesized that the stomatal density of leaves in Mahonia Aquifolium would be greater in the leaves higher up from the ground opposed to the ones lower to the ground. We then predicted that there would be more stomata on the higher leaves because they see significantly more sun than the underlying leaves. Although, this is not what we found. For future research, we would need to measure the distance from the ground to each and every tier of leaves. This would give us a larger field of view on exactly which leaves at which tier had a certain amount of stomata. Instead, what we failed on was only taking data from 3 tiers and then finding the mean of the data on each tier. If we were to stay with Mahonia Aquifolium I don’t believe we would see much of a difference either way. Partially because Mahonia Aquifolium only has a certain number of tiers on the plant which happen to be on the lower side. To continue this research would mean gathering more data altogether. For more accurate results, I would prefer to take images of the microscopic stomata to be able to count more accurately. I believe eyesight counting had a huge effect on our data and is unreliable.
References
Gay, A. P., and R. G. Hurd. “The Influence Of Light On Stomatal Density In The Tomato.” New Phytologist, vol. 75, no. 1, 1975, pp. 37–46., doi:10.1111/j.1469-8137.1975.tb01368.x.
Mott, Keith A., and Odette Michaelson. “Amphistomy as an Adaptation to High Light Intensity in Ambrosia Cordifolia (Compositae).” American Journal of Botany, vol. 78, no. 1, 1991, p. 76., doi:10.2307/2445230.
Grant, Bruce W, and Itzick Vatnick. Teaching Issues and Experiments in Ecology . Vol. 1, Widener University, 2014.
Ocarrigan, Andrew, et al. “Analysis of Gas Exchange, Stomatal Behaviour and Micronutrients Uncovers Dynamic Response and Adaptation of Tomato Plants to Monochromatic Light Treatments.” Plant Physiology and Biochemistry, vol. 82, 2014, pp. 105–115., doi:10.1016/j.plaphy.2014.05.012.
Peñuelas, Josep, and Roser Matamala. “Changes in N and S Leaf Content, Stomatal Density and Specific Leaf Area of 14 Plant Species during the Last Three Centuries of CO2Increase.” Journal of Experimental Botany, vol. 41, no. 9, 1990, pp. 1119–1124., doi:10.1093/jxb/41.9.1119.